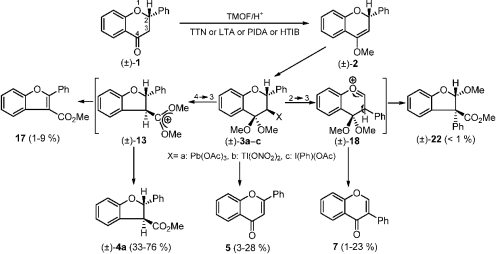 
Therefore, option A is the correct answer. Here, the complex formed is known as anilinium chloride. 
The balanced chemical reaction between aniline and hydrochloric acid is: Due to which the formation of the complex the aniline will dissolve in the solution of hydrochloric acid. In the protonation process, the negative anion from the acid comes and attaches to the positive charge that is present on the amine. Now, the amine is able to undergo the protonation by an acid that is hydrochloric acid. When aniline dissolves in the hydrochloric acid then the lone pair that is present on the amine group will undergo partial delocalisation. Now, the hydrochloric acid as the name tells has acidic nature so when it will react with aniline the acid-base reaction will take place. The molecule is formed when the amine group attaches on the benzene ring. Aniline is an organic compound which has basic nature. In the given question, we have to choose the correct product formed by the reaction between aniline and hydrochloric acid. Hint: For this problem, we have to write the complete reaction of the aniline with hydrochloric acid and after the product is formed we have to study its nature and properties to choose the correct option.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
